Clinical Application
Platform Overview: PRIMABS™-Dx Assay Workflow
PRIMABS-Dx measures baseline mitochondrial priming and ex vivo treatment-induced shift scores using standard clinical formats including flow cytometry and fixed-tissue immunohistochemistry. The platform is designed for seamless integration into existing laboratory infrastructure.
Sample Input
Fresh or viably frozen AML/MM biopsies; fixed solid tumor tissues
PRIMABS Processing
Standardized antibody staining and quantitative detection protocols
Predictive Output
Quantitative priming metrics and actionable treatment recommendation reports
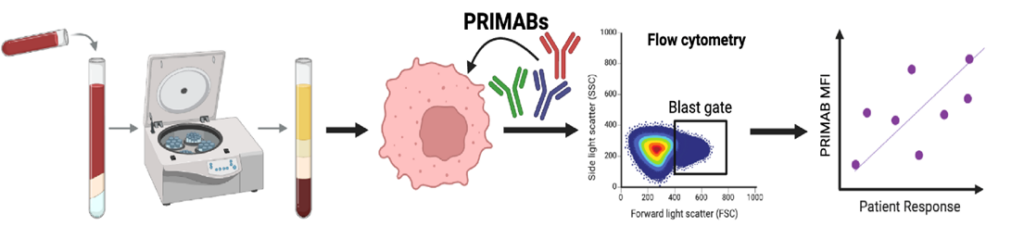
PRIMABs recognized conformational epitopes on intracellular BCL-2 PPIs. Intracellular staining with PRIMABs on Ficoll-purified patient biopsies. Flow cytometry readouts are assessed for correlation to patient response to treatment. Schematic made using BioRender.
Methodology/Advantages
PRIMABS™‑Dx method
Why it matters for clinics and pharma
Measures BCL‑2 family PPIs defining mitochondrial priming using conformation‑specific PRIMABS antibodies in fixed or viably frozen samples
Uses short ex vivo drug exposure followed by priming readout and a “PRIMABS™ Shift Score.
Provides a surrogate functional readout of the final apoptosis checkpoint, downstream of diverse oncogenic pathways.
Works on real‑world biopsies; no fresh tissue requirement; fits standard clinical lab workflows (flow cytometry, IHC).
Supports PD studies, dose/regimen selection, and identification of resistance mechanisms in trials.
Complements genomic profiling, captures convergence of multiple targets on cell‑death commitment.
Application: AML and Multiple Myeloma
Our validated assay platforms deliver actionable insights across key hematologic malignancies, with expansion into solid tumors underway. Each application is designed for real-world clinical implementation and regulatory pathway compatibility.
AML PRIMABS-Dx Flow Assay
Performed on peripheral blood or bone marrow specimens, this assay identifies distinct mitochondrial priming patterns and predicts response to venetoclax-based treatment regimens with high sensitivity and specificity. Turnaround time: 48-72 hours.
Multiple Myeloma Application
Performed on peripheral blood or bone marrow specimens, this assay identifies distinct mitochondrial priming patterns and predicts response to venetoclax-based treatment regimens with high sensitivity and specificity. Turnaround time: 48-72 hours.
