Clinical Applications
Platform Overview: PRIMABS™-Dx Assay Workflow
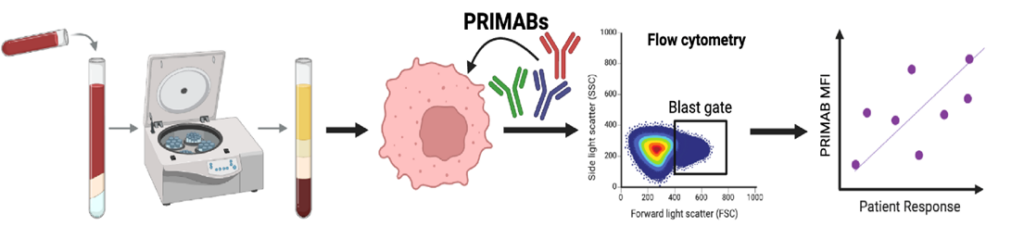
Sample Input
Ficoll purified and fixed peripheral blood or bone marrow biopsies; fixed solid tumor tissues (FFPE)
PRIMABS Processing
Standardized antibody staining and quantitative detection protocols
Predictive Output
Quantitative priming metrics and actionable treatment recommendation reports
Methodology/Advantages
PRIMABS™‑Dx method
Why it matters for clinics and pharma
Measures BCL‑2 family PPIs defining mitochondrial priming using conformation‑specific PRIMABS antibodies in fixed or viably frozen samples
Uses short ex vivo drug exposure followed by priming readout and a “PRIMABS™ Shift Score.
Provides a surrogate functional readout of the final apoptosis checkpoint, downstream of diverse oncogenic pathways.
Works on real‑world biopsies; no fresh tissue requirement; fits standard clinical lab workflows (flow cytometry, IHC).
Supports PD studies, dose/regimen selection, and identification of resistance mechanisms in trials.
Complements genomic profiling, captures convergence of multiple targets on cell‑death commitment.
Clinical Applications: AML and Multiple Myeloma
AML PRIMABS-Dx
Flow cytometry assay on peripheral blood or bone marrow identifies priming patterns and predicts response to venetoclax-based regimens
Myeloma Assay
Identifies biomarker algorithms for standard-of-care combinations with existing and investigational agents
Custom Development
Platform available for custom CLIA assay development with pharmaceutical partners and clinical trial sponsors
PRIMABS™: Scientific Validation

Published, Peer‑reviewed studies at Eutropics have shown that PRIMAB readouts quantitatively correlate with BH3‑mimetic sensitivity and with muktiple targeted and chemo therapies.
